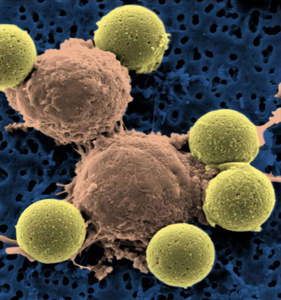
This microscopy image provided by Dr. Carl June on Wednesday, Aug. 10, 2011 shows immune system T-cells, center, binding to beads which cause the cells to divide. Scientists are reporting the first clear success with gene therapy to treat leukemia. (Dr. Carl June/The Associated Press)
Three men who were suffering from advanced chronic lymphocytic leukemia (CLL) are now healthy and cancer-free, thanks to a pair of clinical studies for a promising new gene therapy. The results, announced Wednesday in the New England Journal of Medicine and Science Translational Medicine, have medical researchers in thrall: “I’m getting goosebumps,” says University of Pennsylvania pathologist Michael Kalos, lead author of one of the studies.
Though the sample is small, the findings of Drs. Carl June and Michael Kalos (both of the University of Pennsylvania) indicate the new treatment is better than successful–in addition to eradicating tumors and removing cancer cells from the body for over a year, the reprogrammed T-cells seemingly act as a vaccine, preventing future recurrences of cancer.
The technique works like this: the patient’s T-cells are isolated and genetically reprogrammed using a virus vector that inserts a new gene into their DNA. This new gene prompts the T-cells to create an antibody — known as chimeric antigen receptor or CAR — that specifically targets structures present on the surface of cancer cells.
After the modified T-cells are injected back into the patient, they seek and attach themselves to cancer cells, then obliterate them with a swiftness and efficiency that has researchers astounded. They’ve been dubbed “serial killers,” and each reprogrammed T-cell is responsible for the calculated death of thousands of cancer cells. “Within three weeks the tumours had been blown away, in a way that was much more violent than we ever expected,” said June.
More importantly, however, the “serial killer” cells cause other T-cells to multiply each time they attack, creating more killers with each slain cancer cell. Healthy tissue is left unharmed and the patient’s immune system is not only uncompromised, but bolstered by the additional production of T-cells. “If leukemia does come back, those T-cells (appear to be) armed and ready to eliminate it,” Kalos said.
There’s no reason to suspect that this treatment is only effective against leukemia, either. By modifying the T-cells to target a specific structure on various cancers, the new technique is feasible for treatment of “hard tumor” cancers like breast, lung and prostate. “You can target prostate cancer, for example, by targeting any of the surface molecules that have been shown to be present on prostate cancer,” Kalos says.
These are not the first studies to utilize modified T-cells as cancer treatment, but they are the first to show robust and successful results. The object now is to repeat the process in further studies, which are planned for the coming months.
